Automating the Digital Pathology Workflow
What is Digital Pathology?
Digital pathology is the image-based computer analysis of a biological sample for both clinical and non-clinical applications. Digital pathology is not considered a medical discipline but rather a process or workflow associated with image acquisition, analysis, interpretation, and storage to assist with timely and accurate information. In clinical applications, digital pathology provides a fast and efficient way for healthcare workers to converge on a diagnosis. The digital nature of samples and the analysis process makes it easier for pathologists, histopathologists, oncologists, and other medical experts to share and collaborate. Non-clinical applications include research, education, and biopharmaceutical development. Similar to the clinical side, collaborating with digital images accelerates research timelines and decreases drug development time, especially if we talk about innovative digital imaging solutions such as whole slide imaging.
Whole slide imaging is a critical component of digital pathology workflows, allowing pathologists to scan entire tissue samples at high resolution for detailed analysis and diagnosis. However, whole slide imaging can be time-consuming and labor-intensive, requiring careful handling of delicate tissue samples and precise positioning of the imaging equipment using slide scanners. By automating the handling and positioning of samples and equipment, motion control can significantly improve the efficiency and accuracy of whole slide imaging workflows, ultimately improving patient outcomes and advancing the field of digital pathology. We offer a range of motion control solutions specifically designed for whole slide imaging workflows, including linear stages, nanopositioning stages, and custom motion control systems that help you tackle any automation and accuracy challenges.
While digital pathology instrument architectures will differ between manufacturers and end-use requirements, three overarching sub-systems are the building blocks of any microscopy applications: moving the slide into the camera’s field of view (X and Y Motion), focusing on the sample (Z axis), and managing the optical system integration.
Related Case Studies
Moving the Slide into the Field of View
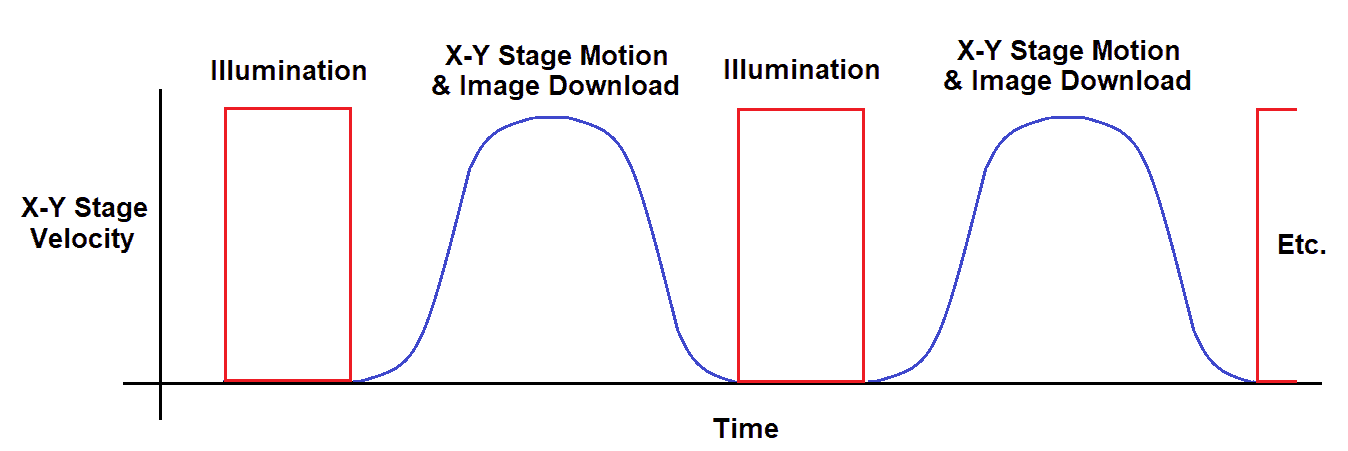
Microscopy design experts turn to Dover Motion when challenged to reduce step-and-settle time and increase overall machine throughput. Our nanopositioning expertise and extensive experience in optics enable us to develop integral, application-ready motion products. For example, the Dover Motion SmartStage™ XY nanopositioning stage can provide the following benefits:
- Reduced settling time:
- Settling ±50 nm in <45 msec after 3mm platform move
- Settling ±50 nm in <15 msec after 333 µm platform move
- Bi-directional repeatability <25 nm
- Sub-micron accuracy
- Holding Stability 5 nm RMS

Constant Velocity Imaging
Constant velocity scanning (sometimes called “fly-over mode”) is the most popular option for high-throughput microscopy and whole slide imaging applications. Most whole slide scanners have input capacities of between 100 and 1,000 slides. The consistent motion of the sample, combined with a synchronized imager, enables high-resolution, high-throughput data collection. As throughput requirements increase especially for slide scanners, more digital pathology platform companies are seeking out the expertise and motion technology provided by Dover Motion. We have made countless constant velocity motion systems for decades. We have invested in the expertise of our staff as well as our product portfolio, which includes both mechanical and control-based solutions. Over the decades, we’ve invested in technologies such as crossed roller guideways, low-noise electronic drive and control, sub-nanometer resolution feedback devices, and advanced electronic triggering capabilities (TOP). Dover Motion first developed Trigger On Position capabilities for demanding semiconductor applications in the late 1980s. We now leverage this approach for microscopy applications that use Time Delay Integration (TDI) cameras. We integrated Trigger On Position (TOP) inside of our SmartStage XY motion controller to provide hardware triggering to line scan cameras based on the actual position of the stage and sample. This provides increased accuracy of the image acquisition during slide scanning. TOP is typically used to trigger cameras and lasers to synchronize them with the motion and position of the stage. For example, combining this software capability, smooth crossed roller bearings, and our ultra-high-performance encoder inside of the SmartStage XY nanopositioning stage enables low spatial triggering jitter and consistent platform movement over the entire travel. As a result, we maintain extremely low-velocity ripple while scanning and ± 30nm stability at rest. Our nanopositioning stage solutions provide the following benefits:
- 5 nm resolution
- Bi-directional repeatability <25 nm
- <10 nm RMS cyclic error
- Camera triggers synchronized to position of the sample with <100 ns
Focusing on the Sample
Positioning the sample under a microscope (X & Y axis) is only one piece of the puzzle. Since no sample is perfectly flat, another important aspect is to dynamically focus the objective for the best quality image. The ability to position the Z-axis assists in obtaining the target optical resolution. The required resolution differs by application. For example, a simple mammalian cell counting application may use a resolution of 2 μm, while digital pathology applications targeting fine structural morphologies of cell nuclei may require 0.35 μm resolution. There are two fundamental methods of focusing in microscopy: contrast and laser autofocusing.
Contrast (Software) Focusing
Contrast focusing is primarily a software-based approach where the image from the camera is analyzed as the Z-axis steps through the nominal and presumed focal planes. Images are then put through a high-pass filter that derives a value for fine-scale contrast. While this method does not require costly autofocusing equipment, the tradeoff is slower focus time. Typically, the Z-axis moves the objective 15 to 30 times before the optimal image can be captured.
Dover Motion created the DOF nanopositioning stage to address the need for a repeatable, quick-to-settle Z-Axis. The DOF provides the following benefits for contrast focusing:
- Fast, accurate, and repeatable 100 nm moves
- 5nm RMS position stability
- 25nm repeatability
- High throughput steps that are quick to settle

Laser Autofocus
Optical-based autofocusing relies on sending a laser diode beam down through the objective where it reflects off the sample or a nearby glass surface. This continuous tracking laser autofocus method provides the highest performance but can be considerably more expensive to implement. This architecture allows the Z-axis stage to be driven by the digital laser autofocus. Dover Motion has been building high-performance, direct drive Z nanopositioing stages for many years with thousands of systems in the field, many of which operate around the clock. Our highest resolution focusing stage is our DOF nanopositioning stage for objective focusing. Applications using the DOF benefit from:
- Fast, accurate, and repeatable 100nm moves
- Integrated optical linear encoder with 1.25nm or 5nm resolution
- Internal, high performance servo drive and control with reduced cabling
- Direct hardware connection to Autofocus systems for easy position control
Managing Optical System Integration
There continues to be a need for many different types of machine architectures to support all conceivable use cases. Often special use cases leverage low-throughput instruments that use techniques such as fluorescence imaging. While processes may vary between platforms and companies, all optical-based instruments are looking for the return of specific light wavelengths. These instruments rely on precision optical filters, commonly on rotary indexers or linear arrays. Often referred to as “filter cubes”, these assemblies contain an excitation filter, an emission filter, and occasionally a beam splitter. Each cube must be quickly and accurately moved into the optical path to pick up the minimal number of photons being emitted by the fluorophore. We have a robust history of working on optical and fluorescence imaging applications. The experts at Dover Motion are consulted on optical architecture options where we work with our customers to design a system with the highest performance and throughput. For example, our expertise provides the following benefits:
- Cost-effective options for both rotary or linear indexing architectures
- Multiple technologies to support a range of price and performance requirements
- Custom design experience for unique filter-changing needs.
The Dover Motion Expertise
Dover Motion provides collaborative solutions for motion control digital pathology platforms so that our customers can focus on what they do best. Our technical experts have a deep understanding of motion control systems, optical imaging systems as well as ancillary application technologies. We understand the environmental impacts of the various systems on the imaging process and can prevent negative effects before they arise. Due to our years of experience in the industry, we are well-versed in automated microscopy, whole slide imaging, control theory, fluorescence microscopy, vibration control, optimizing servo tuning, and structural resonances. Collaborating with our experts enables digital pathology platforms to decrease instrument development time and prevent system challenges before they arise.
Here are some examples of where Dover Motion’s expertise can be of value:
- We consider the impact of other systems, including cables, tubing and temperature control elements and prevent them from hindering the precise platform motion.
- We provide best practices for other critical controlled systems, such as temperature control.
- We normalize the sample plane to the optical beam path, either statistically or dynamically.
- We offer guidance for managing negative system effects (i.e., consequences of isolation).
- We include advanced methods to further reduce step and settle time.
- We support the rotary filter indexer or linear array control.
Building the Right Precision Motion Control System
Dover Motion offers a variety of standard products for your precision motion control needs, and we also specialize in collaborating with our clients to configure the right motion solution for their unique application. With more than 50 years of experience in the life sciences, diagnostics, and factory automation industries, we understand your needs and industry and can provide you with the flexibility you need to hit your cost and schedule targets.
Related Products
The DOF™ microscope stage is optimized for objective focusing with increased travel, higher bandwidth, and lower cost than piezo stages.
| wdt_ID | Travel | 5-9 mm |
|---|---|---|
| 1 | Resolution | 1.25 nm |
| 2 | Repeatability | < 50 nm |
| 3 | Bandwidth | > 225 Hz |
The award winning SmartStage™ XY stage offers high precision and includes an embedded controller inside the stage.
| wdt_ID | Travel | 50 - 200 mm |
|---|---|---|
| 1 | Accuracy | ≤ 10 μm |
| 2 | Repeatability | 0.8 μm |
| 3 | Payload | 10 kg |
SmartStage™ XY Monolithic Stage
The SmartStage XY Monolithic stage, is a linear motor stage in a low-profile XY form factor, with physically integrated controller, designed for precision positioning.
| wdt_ID | Travel | 50 - 75 mm |
|---|---|---|
| 1 | Accuracy | 12 μm |
| 2 | Repeatability | 0.8 μm |
| 3 | Payload (kg) | 10 kg |
XYA™ Linear Stage
The Dover XYA™ series XY linear stage incorporates the motor and controller into the body of the stage. Covers provide pinch point protection during motion.
| wdt_ID | Travel | 100 - 200 mm |
|---|---|---|
| 1 | Accuracy | 50 μm TIR |
| 2 | Repeatability | 5 μm |
| 3 | Payload | 10 kg |
SmartStage™ Shuttle Long Travel Stage
The SmartStage Shuttle is a long travel linear stage with an integrated controller, delivering 5 nm resolution in a compact design for fast, precise movement.
| wdt_ID | Travel | 200 - 500 mm |
|---|---|---|
| 1 | Accuracy | 5 μm |
| 2 | Repeatability | 1.2 μm |
| 3 | Payload | 10 kg |







